In this issue:
Lung Cancer is the leading cause of cancer-related fatalities worldwide. It is estimated at a rate of 1.3 million deaths per year. This study was designed to evaluate the efficacy of photothermal ablation therapy for lung cancer by low-power near-infrared laser and topical injection of indocyanine green (ICG). An 808 nm laser with 250 mW was irradiated for 10 minutes using different dilutions of ICG and the temporal thermal effect was monitored. ICG (1 mL of 0.5 g/L) was heated to a temperature of >30° °C from the base temperature by laser irradiation. The cytotoxic effect of hyperthermia on human lung cancer cells was examined in different temperature and time settings. Cell viability was quantified by both an MTS assay and reculturing. Fatal conditions evaluated by reculturing were as follows: thermal treatment at 55° °C for 5 minutes, 53° °C for 10 minutes, and 51° °C for 15 minutes. The MTS assay study suggested that thermal treatment at 59° °C for 5 minutes and 57° °C for 20 minutes showed a severe cytotoxic effect. In vivo study: nude mouse subcutaneous NCI-H460 human lung cancer xenograft models were used for the study. Saline or 0.5 g/L of ICG was injected topically into the tumor (n=3/group). Tumors were irradiated with a laser at 500 mW for 10 minutes. Although the tumor diameter reached 1 cm within 24 days after treatment in all 3 mice using saline/laser, tumor sizes were gradually reduced in all 3 mice using the ICG/laser. In 2 of the 3 mice using ICG/laser, tumors had disappeared macroscopically. The efficacy of the photothermal ablation therapy by low-power near-infrared laser and a topical injection of ICG was clarified using a mouse subcutaneous a lung cancer xenograft model.
Photothermal Enhancement of ICG
ZD-GNRs were diluted into different concentrations with distilled water. Aliquots of samples were exposed to an
808 nm NIR laser (Laserglow Technologies) for 10 min at varying intensity levels. A laser energy meter was used to measure and calibrate the laser densities. The temperature increase of the solutions was recorded with an infrared thermal imaging system.

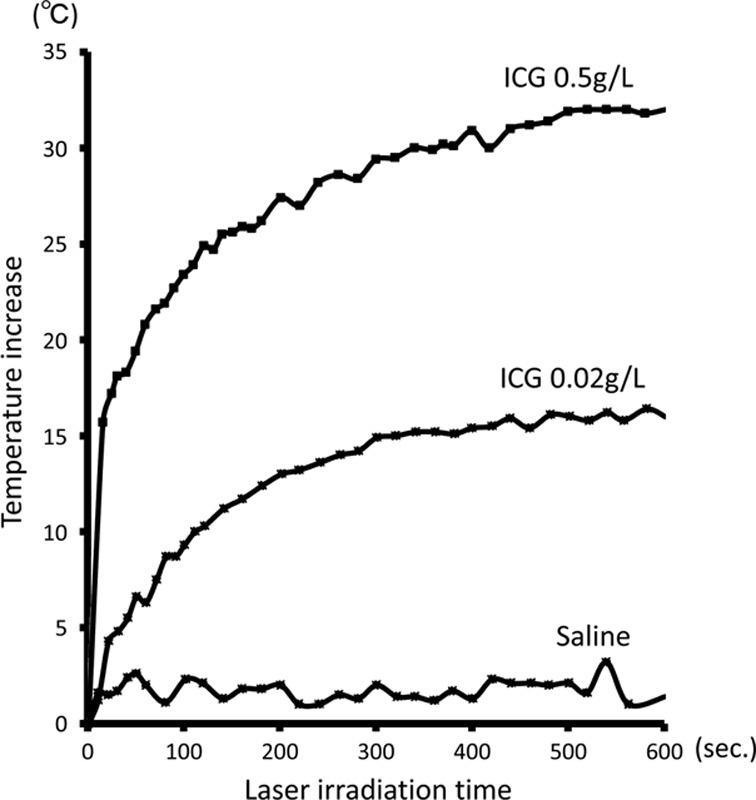
An 808 nm laser with 250 mW output was irradiated using different dilutions of ICG and the temporal thermal effect was monitored. Different dilute concentrations of ICG, 0.5 and 0.02 g/L, and saline were prepared. About 1 mL of these solutions in a conical tube were irradiated with a laser from a distance of 1 cm for 10 minutes. The distance between the tip of the laser fiber and the conical tube was adjusted to the spot size, which can be smaller than the diameter of the conical tube. Our A NIR laser diode with a wavelength of
808 nm was used in this study. A laser power meter console was used in this study. During the irradiation, the surface temperatures of the solutions were monitored using a thermal camera.
There are several limitations to this study. First, in the current study, ICG was administered using a topical injection through the skin into the tumor site. The mouse subcutaneous tumor was then irradiated with the laser from outside the body. To translate this into the clinical treatment of lung cancer, we need to consider how to approach the target lesion. In terms of central lung cancer, we can easily do this using a bronchoscopy and can topically administer ICG under bronchoscopy guidance. This technique, using endoscopy, may potentially be applied to malignant lesions in other organs, such as the esophagus, stomach, and colon. To translate this into the treatment of peripheral lung cancer, one will require precise access to the peripheral lung tumor, which can be achieved using recently developed navigational bronchoscopy.26,27 The second limitation is the limited tissue penetration of NIR light. Kim et al28 have described that NIR light penetrates human tissues to a depth of 10 mm. Tanaka et al29 have also reported that 10 mm is the maximum limit of depth for detecting NIR light. However, taking the thermal conductivity in the ICG-injected tumor into consideration, tumors larger than 1 cm may be treated with this approach. The third limitation is the procedure time. In our experiments, the team used a minimally thin optical fiber (300 μm) that could fit in the accessory channel of the smallest ultrathin bronchoscope (1.2 mm ID); thus, it took 10 to 20 minutes. If the team increase the diameter of the fiber, they can potentially decrease the duration of photothermal ablation treatment.
In conclusion, they clarified the efficacy and feasibility of photothermal ablation therapy by a low-power NIR laser and a topical injection of ICG using a mouse subcutaneous human lung cancer xenograft model. This system may potentially be applied for transbronchial laser ablation of peripheral lung cancers.
Full access to the materials and methodology, can be found by
clicking here.
Details on the 808 nm NIR Laser used in the research can be found by
clicking here.


