In this issue:

Feature: Super-Resolution Microscopy - Advancements in STED
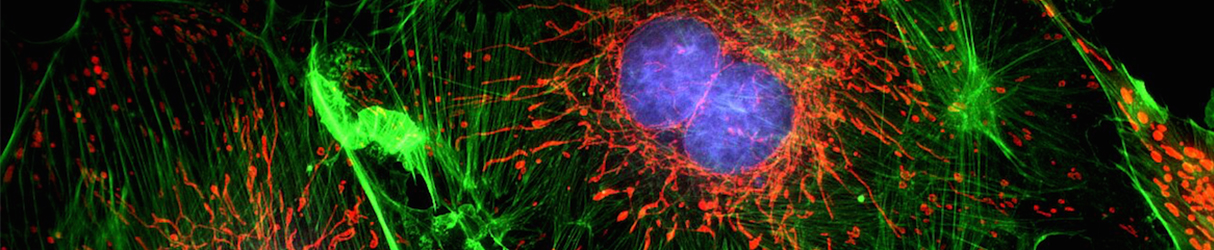
Achieving a spatial resolution that is not limited by the diffraction of light, recent developments of super-resolution fluorescence microscopy techniques allow us to see many biological structures not visible through conventional fluorescence microscopy. New advances in these techniques now give them the ability to image three-dimensional (3D) structures, measure interactions by multicolor colocalization1, and record dynamic processes in living cells at the nanometer scale. It is anticipated that super-resolution fluorescence microscopy will become a widely used tool for cell and tissue imaging to provide previously unobserved details of biological structures and processes.
The advent of super-resolution techniques is partly due to the wave nature of light. The resolution of light microscopy is limited because light is a wave and is subject to diffraction. This diffraction limit of light microscopy, about 200-300 nm in the lateral direction and 500-700 nm in the axial direction, is larger than many subcellular structures, leaving them too small to be observed in detail. Super-resolution techniques aim at overcoming this diffraction barrier, and include techniques that employ nonlinear effects to sharpen the point-spread function (PSF) of the microscope, such as stimulated emission depletion (STED) microscopy.
Other techniques are based on the localization of individual fluorescent molecules, such as stochastic optical reconstruction microscopy (STORM), photoactivated localization microscopy (PALM), and fluorescence photoactivation localization microscopy (FPALM), which we will cover in future editions.
Stimulated emission depletion (STED) microscopy is a widely used super-resolution microscopy technique. It creates super-resolution images by the selective deactivation of fluorophores, minimizing the area of illumination at the focal point, and thus enhancing the resolution for a given system.
In practice, STED uses a second laser (STED or depletion laser) to suppress the fluorescence emission from the fluorophores that are located off the center of the excitation. This effect by itself does not give super-resolution, but the trick is to focus the STED laser light into a ring shape - commonly called a donut - around the focus where the fluorophores have been excited. In this fashion, only the fluorophores in the outer region of the focus are de-excited, leaving an effectively smaller spot from which fluorescence can emanate.
The efficiency of depletion of excited molecules, and thus the STED performance, depends greatly on the properties of the STED laser. Key parameters are optical output power, pulse width and repetition rate, which must be matched and synchronized to the repetition rate of the excitation laser. NIR range lasers with pulse width of several hundred picoseconds are ideal for obtaining high STED resolution, and minimize photobleaching.
There have been many exciting developments in research through STED techniques. STED combined with mirror-enhanced, axial narrowing, super-resolution (MEANS) microscopy improves axial resolution six-fold and lateral resolution two-fold for STED nanoscopy. A research team in Germany recently refined STED by modifying image acquisition in a way that suppresses background efficiently. The resulting enhanced image quality is well suited for quantitative data analysis of 3D molecules and cell structures. Finally, an optical fiber based version of STED developed at Boston University (and presented at CLEO 2016) shows how excitation and depletion light can exist in the same fiber and how this could greatly improve the stability of the STED system.
Laserglow Technologies offers many laser products - both CW and pulsed, that are ideal for STED microscopy research. We look forward to helping our customers reach the next stage in their microscopy research project.
Terms:
1 - In fluorescence microscopy, colocalization refers to observation of the spatial overlap between two (or more) different fluorescent labels, each having a separate emission wavelength, to see if the different "targets" are located in the same area of the cell or very near to one another.
Sources:
1. Super resolution fluorescence microscopy, Huang et. al., National Institutes of Health - US Public Library of Medicines, NIHMSID: NIHMS179491 NIH Article
2. STED Microscopy Made Easy, Schulz & Koenig- Photonics.com, Dec 2014 Photonics Dec 2014
3. Mirror approach boosts super-resolution microscopy for cell studies, BioOptics World, June 2016 BioOptics World Editorial
4. Superresolution Microscopy: All-fiber illumination brings superior stability to STED - BioOptics World, July 2016 BioOptics World Editorial
Laserglow in Research: Optogenetic Investigation of Sleep - Driving Neurons
Laserglow's LRD-0470 Cyanea™ Series Collimated Diode Laser was recently used by a research group to study the role of neurons containing melanin-concentrating hormone (MCH) in driving sleep. The study was a collaboration between researchers from University of South Carolina, Yale University and the Veterans Administration Medical Center, Charleston, South Carolina. The blue 470 nm diode laser was used to activate Channelrhodopsin-2 (ChR2), the main plasmid used in this research.
Neurons containing melanin-concentrating hormone (MCH) are located in the hypothalamus. In mice, optogenetic activation of the MCH neurons induces both non-rapid eye movement (NREM) and rapid eye movement (REM) sleep at night, which is the normal wake-active period for nocturnal rodents. In this study, the researchers selectively activated these neurons in other mammal models to test the validity of the sleep network hypothesis in other species.
Channelrhodopsin-2 (ChR2) driven by the MCH promoter was selectively expressed by MCH neurons after injection of viral vector / plasmid into the hypothalamus of Long-Evans rat models. An in vitro study confirmed that the optogenetic activation of MCH neurons faithfully triggered action potentials.
In a second in-vivo study, the viral and control vectors were delivered into the hypothalamus. Three weeks later, baseline sleep was recorded for 48 h without optogenetic stimulation (0 Hz). Subsequently, at the start of the lights-off cycle, the MCH neurons were stimulated at 5, 10, or 30 Hz for 24 h. Sleep was recorded during the 24-h stimulation period. Optogenetic activation of MCH neurons increased both REM and NREM sleep at night, whereas during the day cycle, only REM sleep was increased. Delta power, an indicator of sleep intensity, was also increased.
In control rat models without ChR2, optogenetic stimulation did not increase sleep or delta power. The researchers state that these results lend further support to the view that sleep-active MCH neurons contribute to drive sleep in mammals. Read the full paper at: PubMed Full Paper
News: 'Flying saucer' quantum dots hold secret to brighter, better lasers
Fresh insights into living cells, brighter video projectors and more accurate medical tests are just three of the innovations that could result from a new way of fabricating lasers. The new method, developed by an international research team from University of Toronto Engineering, Vanderbilt University, the Los Alamos National Laboratory and others, produces continuous laser light that is brighter, less expensive and more tunable than current devices, by using nanoparticles known as quantum dots.
By carefully controlling the size of the quantum dots, the researchers can 'tune' the frequency, or color, of the emitted light to any desired value. By contrast, most commercial lasers are limited to one specific frequency, or a very small range, defined by the materials they are made from. This flexible tuning would give a boost to scientists looking to study diseases at the level of tissues or individual cells by offering new tools to probe biochemical reactions. They could also enable laser display projectors that would be brighter and more energy efficient than current LCD technology.
The ability of quantum dots to produce laser light has been limited so far, because the amount of power required to excite the quantum dots to that level is very high. The team overcame this problem by changing the shape of the quantum dots, rather than their size. They were able to create quantum dots with a spherical core and a shell shaped like a Skittle, an M & M or a flying saucer - a 'squashed' spherical shape known as an oblate spheroid.
The mismatch between the shape of the core and the shell introduces a tension that affects the electronic states of the quantum dot, lowering the amount of energy needed to trigger the laser. As reported in a paper published in Nature, the innovation means that the quantum dots are no longer in danger of overheating, so the resulting laser can fire continuously. The team hopes that in the future they would be able to excite quantum dots with electricity instead of light, with output scaling up to miliwatts and even watts, which would be an important breakthrough for laser development. Read more at: Phys.org full article