
Feature: Super-Resolution Microscopy - Advancements in STED
Technology plays a vital role in understanding the complexity of the human immune system. Procedures that were extremely manual and time intensive, now take a fraction of the time. One such technique is Flow Cytometry that investigators use to diagnose health conditions, especially those related to the immune system.
Cytometry is the quantitative analysis of cells and cell systems - measuring things like cell size, number of cells, cell shape and structure, types of proteins etc. The next time your doctors does a blood count, it was probably done using cytometry. Similarly, cytometry is also used in cell biology research and in medical diagnostics to characterize cells in a wide range of applications associated with diseases such as cancer and AIDS.
Components of a Flow Cytometer
Three major components make up a Flow Cytometer - a fluidic system that can deliver individual cells in a fluid stream that can be analyzed; an optical system that is comprised of lasers, mirrors, and filters to analyze the cells in the fluid stream; and an electronic system that converts the results into electronic signals.
The Fluidic System
Flow cytometry uses a fluid suspension, commonly known as sheath fluid, to analyze individual cells and simultaneously measure multiple parameters of each individual cell. Thousands of cells per second can be analyzed as they pass through the liquid suspension. This allows researchers to generate large data sets from samples can be small and limiting.
The fluidic system refers to a flow of a cell or particle through a sensing volume to obtain serial analysis of particles on a one by one basis. There are three primary purposes of the fluidics in a flow cytometer. The first purpose is to deliver a sample containing the suspended cells or particles to the flow cytometer. Secondly, the fluidics allows for positioning the sample stream ensuring only one particle or cell is measured at a time. And lastly, the fluidics allows to efficiently sort and collect particles of interest based on the results of the particle analysis.
Sheath fluid is the fluidic suspension that flows into a flow cytometer. Upon reaching laminar flow, the sample cells or particles are injected into the stream at a higher pressure. Hydrodynamics ensures that the cells line up in single file in the direction of the flow of the sheath fluid. (4) Different formulations of sheath fluid are used by researchers, depending on the experimental needs. Sometimes dyes are added to cell samples that bind to the cellular components. These "tagged"cells fluoresce when specific wavelength laser light is shone at it. The fluorescence emissions can then be spectrally filtered.
The Optical System
The purpose of the optical system is to illuminate the cells or particles present in the sheath fluid using lasers. Lasers are used in flow cytometers because of two main reasons - lasers are coherent and monochromatic sources of light. This means that a very narrow wavelength can be specified and the light output will always be in phase. These two properties of lasers are critical for flow cytometry. The sheath fluid flows at a high stream velocity, therefore it is critical that the laser illumination spot be very small and focused, to illuminate a cell or particle.
Another reason for using lasers is that their output power, which essentially measures of how many photons are output per unit time, is usually adjustable. Flow cytometer lasers typical range in power from 20mW to 100mW. plasma allowing for smaller footprint of cytometers, low power requirements, and short warm-up periods.
The most common wavelengths of lasers used in flow cytometry are: 355 nm, 375 nm, 405 nm, 488 nm, 532 nm, 561 nm, and 640 nm. The lasing medium in these lasers is solid, as opposed to a gas or plasma allowing for smaller footprint of cytometers, low power requirements, and short warm-up periods.
When the laser of a specific wavelengths strikes a cell, the light is either reflected by the cell or it is absorbed. Depending of the response to the laser, various properties of the cell can be analyzed. Laser light reflected from a cell can either follow a forward scatter or a side scatter. Forward scattered (FSC) light is refracted by a cell in the same direction that the light was originally traveling. Side scattered (SSC) light is refracted by cells in a direction that is different from the original light path
If a cell has fluorescent properties or is "tagged"with fluorescent dyes, when laser light is shown on them, they absorb the energy of the light and "fluoresce"at a higher wavelength. E.g. fluorescein isothiocyanate (FITC) is a dye that efficiently absorbs light from a blue (488nm) laser and produces a green (525nm) emission while phycoerythrin (PE) also absorbs light from a blue (488nm) laser but produces an orange (575nm) emission.
The Electronic System
The reflected or absorbed emissions can be optically separated by a series of filters, mirrors and beam splitters. Forward scattered light is typically used to determine the size of a cell or particle. Larger the cell or particle, larger the forward scatter.
Side-scattered light is usually used to determine the granularity and complexity of the cell in the light path. E.g. highly granular cells with a large amount of internal complexity, like neutrophils, will produce more side-scattered light, and a higher side-scatter signal than cells with a low-granularity and complexity.
Fluorescent emissions are typically used to determine if a range of fluorescent markers are present or This optically separated light is then directed to photo detectors that converts the light into electronic signals, at a rate as fast as 5000 cells /sec, allowing for the collection of cell specific data including FSC, SSC and the range of chosen fluorescent emissions. This data is then usually presented in the form of cytograms (dot plots) for analysis.
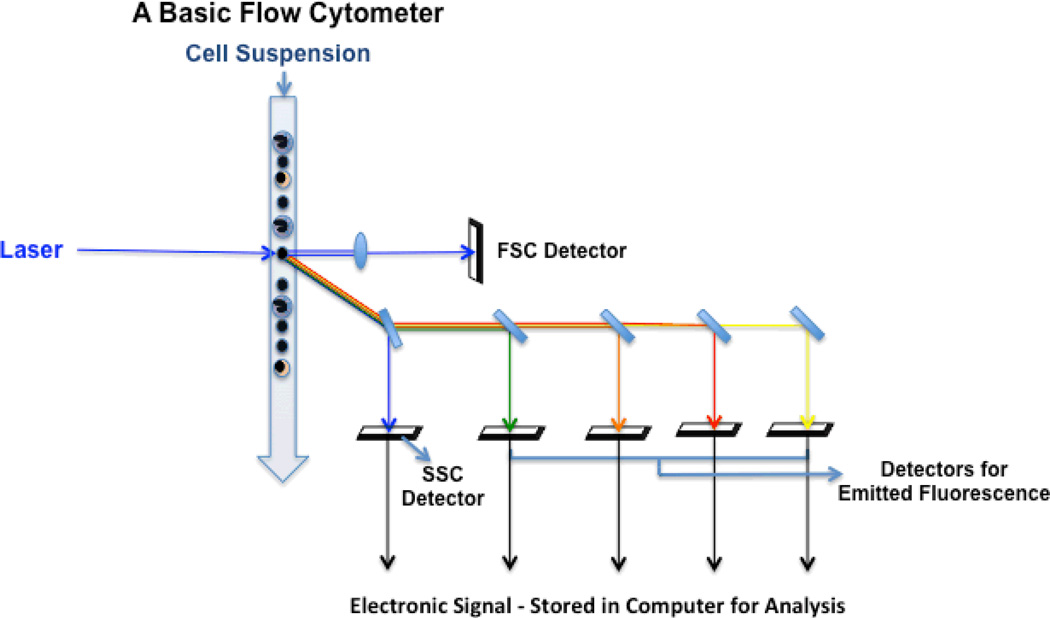
Illustrated are the basic elements of a simple flow cytometer. This includes a laser light source used to interrogate the cells delivered via a stream of fluid. Reflected laser light or fluorescence emissions are collected using a series of mirrors (blue rectangles) and lenses (blue ovals) to detectors that convert light signals to electronic signatures that can be processed and stored. Source - https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3636160/figure/F1/)
Sources